Regulations on Waste Classification According to the Ministry of Health
Medical waste management is one of the crucial legal requirements promulgated by the Ministry of Health to ensure environmental safety, as well as the health and safety of patients, medical staff, and the community. Proper classification of medical waste and the application of appropriate medical waste treatment regulations are mandatory legal obligations for all healthcare facilities. This aims to protect the environment, control the risk of infection, support standardized medical waste treatment systems, and reduce the risk of pathogen dispersion into the community.
1. Overview of Medical Waste
In medical examination, treatment, healthcare, and medical research activities, environmental issues are always a major challenge alongside the mission of saving lives. Medical waste is not simply ordinary garbage. It contains potential hazards capable of spreading diseases and polluting soil, water, and air if not strictly controlled. Especially in the context of increasingly complex emerging infectious diseases, medical waste management has become a paramount biosafety shield.
According to Circular 20/2021/TT-BYT, medical waste is waste generated from the activities of healthcare facilities, divided into two main groups: Hazardous medical waste and ordinary medical waste. Hazardous medical waste is waste containing hazardous elements, including infectious pathogens, toxic chemicals, cytotoxic substances, radioactive materials, or sharps, which can directly or indirectly pose a danger to human health and the environment. Within this, hazardous medical waste includes infectious waste and non-infectious hazardous waste.
Ordinary medical waste includes ordinary solid waste (domestic and administrative waste not contaminated with pathogens or hazardous chemicals) and non-hazardous liquid waste (such as medical wastewater before centralized treatment). Clear segregation right at the source of generation is the foundation for applying different collection and treatment procedures, optimizing costs, and minimizing the risk of infection or environmental pollution.
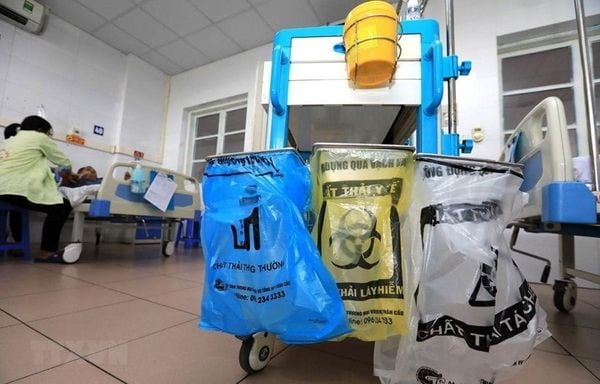
Medical waste is classified in detail by plastic bag color: ordinary waste (green), infectious waste (yellow), recyclable waste (white).
2. Classification of Medical Waste According to Ministry of Health Regulations
According to Article 6 of Circular 20/2021/TT-BYT, classification must adhere to the following principle: Medical waste must be classified right at the place and time of generation. Each type of medical waste must be separately classified into packaging, tools, and waste storage equipment as prescribed in Article 5 of this Circular.
-
In cases where hazardous medical wastes are not capable of reacting or interacting with each other and are subject to the same treatment method, they may be classified together in the same packaging, tool, or storage equipment (except for infectious sharps).
-
If infectious waste is mixed with other wastes, the waste mixture must be collected, stored, and treated as infectious waste, and continue to be managed according to the properties of the waste post-treatment.
-
Regarding the placement of waste classification packaging and tools: At departments, rooms, and units, suitable and safe locations must be arranged to place packaging, tools, and storage equipment for medical waste classification; At these locations, there must be instructions on how to classify and collect the waste.
2.1. Classification of Infectious Waste
This is a group of waste containing pathogens (bacteria, viruses, parasites, fungi) capable of infecting humans. By regulation, this group is subdivided into the following types to implement the most appropriate medical waste treatment plan:

Regulations on the color of medical waste bins.
-
Infectious sharps waste: Includes syringes, syringes with attached needles, sharp ends of IV tubing, puncture needles, acupuncture needles, scalpel blades, nails, surgical saws, broken glass pieces, and other used sharp objects discarded with body blood or containing pathogenic microorganisms. This is the most dangerous medical waste as it can cause physical injury (punctures, cuts) and directly introduce pathogens into the bloodstream. By regulation, infectious sharps must be disposed of in puncture-resistant bins or boxes that are yellow.
-
Non-sharp infectious waste: Includes cotton, bandages, gauze, gloves, and other non-sharp wastes that are soaked, stained, or contain body blood or pathogenic microorganisms; discarded vials of inactivated or attenuated vaccines; liquid infectious waste (including drainage fluid after surgery, medical procedures, discarded fluids containing human blood or pathogenic microorganisms). This is the most frequently generated medical waste during daily patient care. By regulation, non-sharp infectious waste must be placed in a yellow bin lined with a bag.
-
High-risk infectious waste: Includes specimens, containers, and items contaminated with specimens, and discarded specimen-contaminated waste from laboratories of biosafety level II or higher; waste generated from isolation wards, isolation treatment areas, and sampling areas for patients with group A and group B dangerous infectious diseases. This waste group usually appears in laboratories and epidemic treatment areas. By regulation, high-risk infectious waste must be placed in a yellow bin lined with a bag.
-
Anatomical waste: Includes discarded human tissues, body parts, and experimental animal carcasses. This is a specific waste group related to spiritual and ethical issues; therefore, it is often required to be destroyed by incineration or burial according to a separate procedure. By regulation, anatomical waste must be double-bagged or placed in a yellow bin lined with a bag.
2.2. Classification of Non-Infectious Hazardous Waste
The non-infectious hazardous medical waste group is identified by chemical factors and toxicity higher than prescribed safety levels, including toxic chemicals generated from diagnosis and treatment activities. By regulation, this group is subdivided as follows:
-
Discarded chemicals with hazardous components/properties exceeding the hazardous waste threshold or with hazard warnings on the manufacturer's packaging.
-
Discarded pharmaceuticals belonging to the cytotoxic group or with hazard warnings on the manufacturer's packaging.
-
Bottles, vials containing drugs or chemicals, and tools contaminated with drugs or chemicals belonging to the cytotoxic group or with hazard warnings.
-
Broken, damaged, or used discarded medical equipment containing mercury, cadmium (Cd); discarded batteries and accumulators; discarded lead-coated materials used for radiation shielding.
-
X-ray film washing solutions, wastewater from testing and analysis equipment, and discarded solutions with hazardous elements exceeding the hazardous waste threshold.
-
Other medical waste with hazardous components/properties exceeding thresholds or with manufacturer hazard warnings.
For classification, non-infectious hazardous waste must be classified by hazardous waste code to be stored in appropriate packaging, tools, and storage equipment. Common packaging can be used for hazardous wastes with the same properties, incapable of reacting or interacting, and treatable by the same method.
-
Solid non-infectious hazardous waste must be contained in bags or bins, or bag-lined bins that are black.
-
Liquid non-infectious hazardous waste must be contained in tightly capped liquid storage containers, labeled with the code and name of the stored waste.
2.3. Ordinary Solid Waste
This is a group of medical waste that does not fall under hazardous or infectious medical waste. Ordinary solid waste is generally collected and treated similarly to domestic waste at normal facilities, but it still needs to be stored and monitored to avoid mixing with hazardous medical waste. This group includes:
-
Domestic solid waste generated from the daily activities of medical staff, patients, patients' families, trainees, visitors, and outdoor waste in healthcare facilities (except domestic waste generated from isolation and treatment areas for dangerous infectious diseases).
-
Discarded chemicals without hazardous components/properties exceeding thresholds.
-
Bottles, vials containing drugs/chemicals, tools contaminated with drugs/chemicals not in the cytotoxic group or without hazard warnings; discarded vaccine vials not belonging to the inactivated or attenuated types.
-
Non-infectious sharps without hazardous components exceeding thresholds; infectious waste after being treated to meet national environmental technical regulations.
-
Sludge from wastewater treatment systems without hazardous properties exceeding thresholds; ash and slag from solid medical waste incinerators without hazardous properties exceeding thresholds.
Regarding the classification of ordinary solid waste:
-
Ordinary solid waste not used for recycling must be contained in bags or bins, or bag-lined bins that are green. Sharp waste must be placed in puncture-resistant containers.
-
Ordinary solid waste used for recycling must be contained in bags or bins, or bag-lined bins that are white.
2.4. Non-Hazardous Liquid Waste
Non-hazardous liquid waste includes discarded drug solutions and chemicals not in the cytotoxic group, without manufacturer hazard warnings, not containing hazardous elements exceeding thresholds, and not containing pathogenic microorganisms. Regarding classification, non-hazardous liquid waste must be contained in tightly capped liquid waste containers, labeled with the name of the stored waste.
3. Medical Waste Collection and Treatment Procedures
After clearly identifying and classifying each group of medical waste, the next step is to carry out collection and storage according to the correct procedures to limit the risk of infection and environmental pollution. Circular 20/2021/TT-BYT clearly stipulates that this task must be carried out at the place of waste generation immediately as it arises and before moving to the storage area.

Medical waste groups are classified and stored separately.
For infectious medical waste:
-
Infectious waste must be collected separately from the point of generation and transferred to a temporary storage area within the healthcare facility; before collection, bags must be tightly tied, and bins must have tight lids to prevent fluid leakage.
-
For high-risk infectious waste, if the facility has on-site disinfection equipment, it should be pre-treated near the point of generation to eliminate pathogens before transport; if not, this waste must be tightly sealed in double bags before being placed in a special bin labeled "HIGH-RISK INFECTIOUS WASTE".
-
Liquid infectious waste is managed through the medical wastewater collection system and treated according to general medical wastewater regulations before discharge into the environment.
-
The collection frequency for infectious waste is at least once a day for healthcare facilities, and at least once a month for infectious sharps if the generated amount is less than 5 kg/day.
For non-infectious hazardous medical waste:
-
Collected and stored separately at the waste storage area within the healthcare facility.
-
Broken, damaged, or used medical equipment containing mercury is collected and stored separately in plastic boxes or suitable materials, ensuring no leakage or dispersion of mercury vapor into the environment.
For ordinary solid waste:
-
Recyclable ordinary solid waste such as paper, plastic, metal, and clean glass must be collected separately into specialized tools, bins, or bags, with clear identification symbols to serve recycling and reuse activities according to environmental protection regulations.
-
Non-recyclable or ineligible ordinary solid waste, including domestic waste mixed with impurities, dirty packaging, and unclassifiable materials, must be collected into a separate bin system and handed over to collection, transport, and treatment units according to domestic solid waste management procedures.
-
Separating these two groups aims to limit mixing recyclable waste with waste requiring landfilling or incineration, while reducing the final volume of waste to be treated.
For non-hazardous liquid waste:
-
Must be collected into the centralized wastewater collection and treatment system of the healthcare facility, managed according to medical wastewater regulations, ensuring treatment meets environmental standards before discharge. Managing non-hazardous liquid waste according to medical wastewater management procedures helps control the risk of water source pollution while ensuring compliance with legal regulations on environmental protection and biosafety in medical activities.
Classifying medical waste right at the source in accordance with Ministry of Health regulations is a key step that determines the effectiveness of the entire collection, storage, and treatment process. Separating infectious waste, non-infectious hazardous waste, ordinary solid waste, and non-hazardous liquid waste not only helps prevent the risk of spreading pathogens, ensuring safety for medical staff and patients, but also contributes to protecting the environment, enhancing management efficiency, and ensuring legal compliance in medical operations.
References:
-
Thư viện pháp luật (Law Library), Circular 20/2021/TT-BYT regulating medical waste management within the premises of healthcare facilities, accessed at: https://thuvienphapluat.vn/van-ban/Tai-nguyen-Moi-truong/Thong-tu-20-2021-TT-BYT-quan-ly-chat-thai-y-te-trong-pham-vi-khuon-vien-co-so-y-te-496061.aspx
-
Ministry of Health Web Portal, Circular No. 20/2021/TT-BYT detailing what you need to know about medical waste classification and collection, accessed at: https://moh.gov.vn/chuong-trinh-muc-tieu-quoc-gia/-/asset_publisher/7ng11fEWgASC/content/thong-tu-so-20-2021-tt-byt-quy-inh-chi-tiet-viec-phan-loai-thu-gom-chat-thai-y-te-can-biet
Comments:
There are no comment for this news.
 Tiếng anh
Tiếng anh  Vietnam
Vietnam 






.png)
.png)

